Product Specification Sheets
Are product spec sheets created manually or in independent systems?
Customers of chemical companies require product spec sheets to assist them in the manufacture of their products. This data often includes results of formula lab analysis, hazard information, and other operating data. The goal is to provide as much information as the customer needs to use the product safely and efficiently in their own manufacturing process. VicinityChem brings data from formula management, quality, and laboratory analysis in one place, streamlining the effort to generate the product spec sheets.
• Utilizes centralized data, creating one source of data rather than separate silos of information
• Supports multiple formats, becoming agile to various customers or products family requirements
• Generate documents from any screen in the system so any user can author a document with access rights
• Publish static documents for users to draw from after they are approved internally
REACH Compliance
How are you currently handling the European standards to ship products into that region?
The purpose of REACH is to limit the human and environmental risks posed by using certain chemicals in the manufacturing process. All chemicals entering the EU must not use specific Substances of Very High Concern (SVHC) and phase them out of their manufacturing process. While many companies may not ship into the EU today, many other regions are adopting similar requirements. Additionally, a non-EU company may use a product that is then shipped into the EU. If a company wants to ship into the EU or sell to a company that ships into the EU, REACH compliance becomes an important consideration.
• Flag chemicals as SVHC in a central database
• Calculate the existence or concentration of SVHC chemical in a produced item or formula
• Disclose the non-existence of SVHC in the product via the European Chemicals Agency website
• Utilize the VicinityChem where-used and component substitution to identify and eliminate SVHC chemicals from formulas
Customer Certificates of Analysis
Are your lab personnel manually creating COAs for customers and do you struggle to manage different customer driven formats?
Certificates of Analysis have become more than a technical document issued by the quality department. They have become a customer service tool used to differentiate chemical manufactures. More customers are requiring more sophisticated and tailored COAs based on their requirements. Suppliers are having to comply with these or potentially lose the business. VicinityChem provides a way to manage multiple formats of the COA, supports multiple specifications, and streamlines the process of creating the document.
• Customizable COA formats in Crystal Reports or Microsoft SSRS
• Generate COAs from shipping with data being controlled by quality
• Track multiple specifications for a formula or chemical allowing some tests to be run for some customers and different tests for others
• Distribute COAs via email, print, or self-serve on the web
SDS/GHS
How are your SDS documents generated today? Is it a combination of formulas and Word or Excel? Have you thought about streamlining this process?
Most chemical companies have a requirement to generate an SDS or other product-specific safety documents. For some companies, this is fairly basic because their products are narrow in hazard scope while others find it much more complex. The creation and distribution of these documents can be a very laborious task for most companies. VicinityChem can either render an SDS from centralized data or it can integrate with more robust authoring tools to streamline the process.
- Maintain formula, material specifications and hazard information centrally
- Author and distribute SDS documents from any sales order shipping, formula, item, or batch screen in the system
- Integrate formula and chemical data with external SDS authoring packages for complex safety product lines
- Distribute one or many SDS documents in a batch to speed the process
- Archive SDS documents so they can be hosted in the website or require regulatory approval before distribution
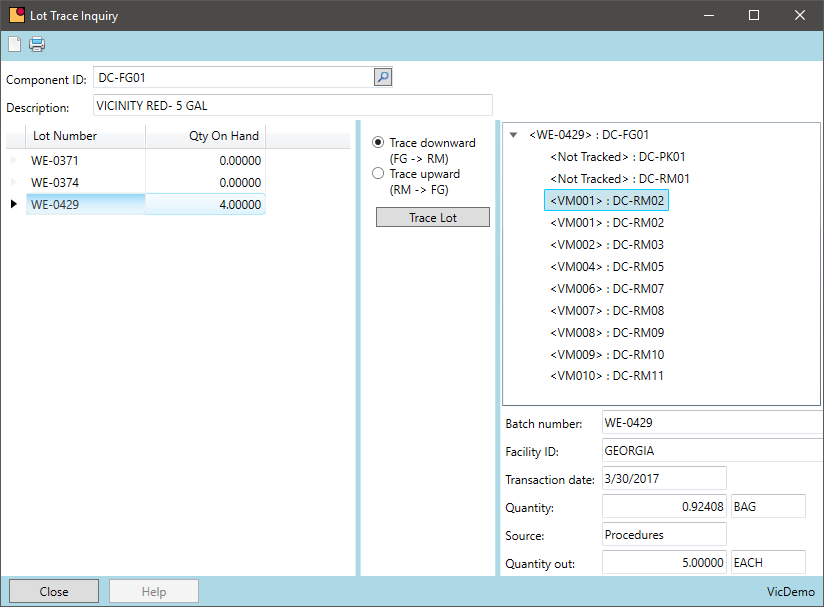
Lot Recall
What steps would you go through to recall a product or track a lot through your existing process?
At some point, most chemical companies face a lot recall for audit purposes or for a live recall of a product. This can be a very manual process for some companies tracing through manual batch tickets or combing through inventory transactions with no way to easily navigate the lot path. VicinityChem makes this process very simple. From raw material through intermediates to finished goods VicinityChem makes a lot trace as simple as pressing a button.
- Lot trace any inventory item — purchased or manufactured
- Assign lots for inbound chemical at receiving and use them on a batch or sell them
- Consume chemical lots in a batch to make one or more output items resulting in a lot tracked item
- Trace unlimited levels of intermediates with no additional work
- A simple user interface to trace from input raw materials, through intermediates to the produced finished goods. This can also be done in reverse
- Generate a custom lot trace report to send to auditors, customers, or suppliers
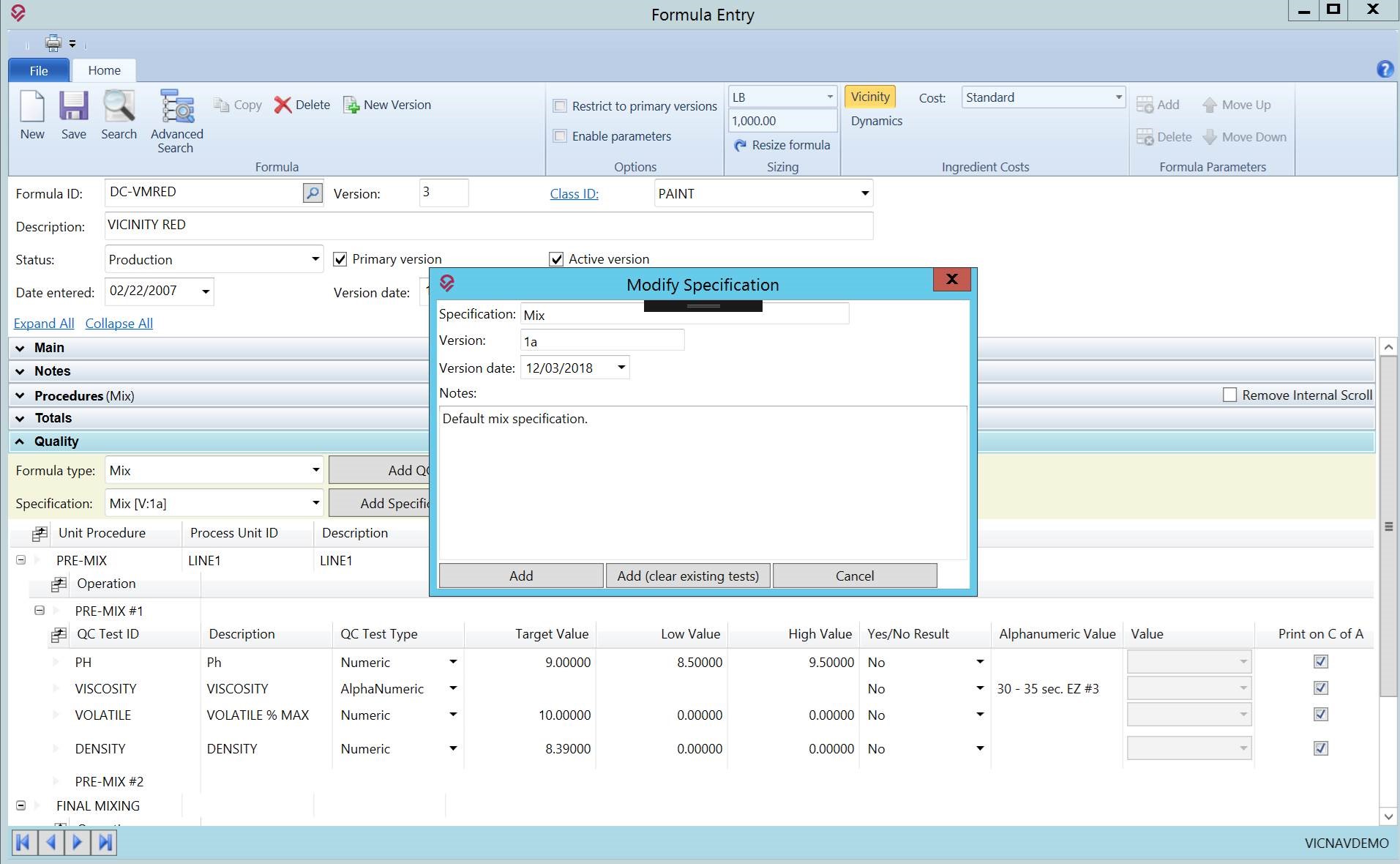
Track Product Specifications
Can your system track various specifications for a produced item?
Chemical manufacturers need to have quick access to all impacted formulas in the event of limited ingredient availability, investigating supplier issues, or evaluating costing impacts on overall production. A formula management software like VicinityChem helps users identify formulas that need to be reviewed.
- Ability to store multiple product specifications against a formula or item
- Record results against one or more specifications to ensure all required tests are being performed
- Select one or more specifications to test a batch or a lot
- Print COAs using a single specification
- Search available lots based on a specification